ISO 11137-2:2013
Sterilization of health care products - Radiation - Part 2 : establishing the sterilization dose
ISO 11137-2:2013 specifies methods for determining the minimum dose needed to achieve a specified requirement for sterility and methods to substantiate the use of 25 kGy or 15 kGy as the sterilization dose to achieve a sterility assurance level, SAL, of 10−6. ISO 11137-2:2013 also specifies methods of sterilization dose audit used to demonstrate the continued effectiveness of the sterilization dose.ISO 11137-2:2013 defines product families for sterilization dose establishment and sterilization dose audit.
ISO 11137-2:2013 specifies methods for determining the minimum dose needed to achieve a specified requirement for sterility and methods to substantiate the use of 25 kGy or 15 kGy as the sterilization dose to achieve a sterility assurance level, SAL, of 10−6. ISO 11137-2:2013 also specifies methods of sterilization dose audit used to demonstrate the continued effectiveness of the sterilization dose.
ISO 11137-2:2013 defines product families for sterilization dose establishment and sterilization dose audit.
ISO 11137-2:2011 specifies methods of determining the minimum dose needed to achieve a specified requirement for sterility and methods to substantiate the use of 25 kGy or 15 kGy as the sterilization dose to achieve a sterility assurance level, SAL, of 10-6. ISO 11137-2:2011 also specifies methods of sterilization dose audit used to demonstrate the continued effectiveness of the sterilization dose. ISO 11137-2:2011 defines product families for sterilization dose establishment and sterilization dose audit.
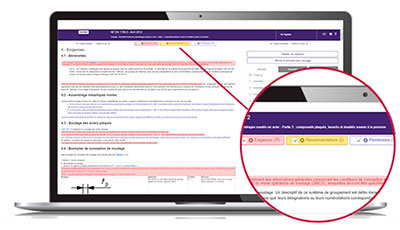
The Requirements department helps you quickly locate within the normative text:
- mandatory clauses to satisfy,
- non-essential but useful clauses to know, such as permissions and recommendations.
The identification of these types of clauses is based on the document “ISO / IEC Directives, Part 2 - Principles and rules of structure and drafting of ISO documents ”as well as on a constantly enriched list of verbal forms.
With Requirements, quickly access the main part of the normative text!
At a glance, you will be able to identify the additions, deletions or modifications to a text, table, figure and formula.
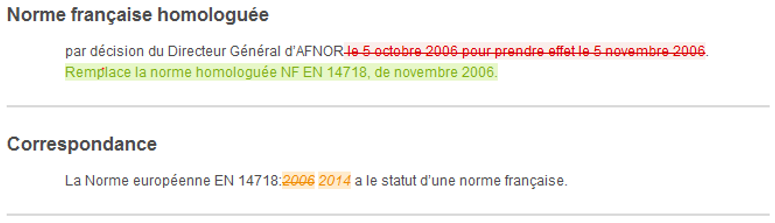
The Redlines + service is offered to you on the collection of French standards in force, in French language and in HTML and PDF format.
For an overview of the service, click on View a standard in redline format
COBAZ is the simple and effective solution to meet the normative needs related to your activity, in France and abroad.
Available by subscription, CObaz is THE modular solution to compose according to your needs today and tomorrow. Quickly discover CObaz!
Request your free, no-obligation live demo
I discover COBAZ