ISO 20776-2:2021
Clinical laboratory testing and in vitro diagnostic test systems - Susceptibility testing of infectious agents and evaluation of performance of antimicrobial susceptibility test devices - Part 2: Evaluation of performance of antimicrobial susceptibility test devices against reference broth micro-dilution
This document establishes acceptable performance criteria for antimicrobial susceptibility test (AST) devices that are used to determine minimum inhibitory concentrations (MIC) of bacteria to antimicrobial agents in medical laboratories.This document specifies requirements for AST devices and procedures for assessing performance of such devices. It defines how a performance evaluation of an AST device is to be conducted.This document has been developed to guide manufacturers in the conduct of performance evaluation studies.
This document establishes acceptable performance criteria for antimicrobial susceptibility test (AST) devices that are used to determine minimum inhibitory concentrations (MIC) of bacteria to antimicrobial agents in medical laboratories.
This document specifies requirements for AST devices and procedures for assessing performance of such devices. It defines how a performance evaluation of an AST device is to be conducted.
This document has been developed to guide manufacturers in the conduct of performance evaluation studies.
ISO 20776-2:2007 establishes acceptable performance criteria for antimicrobial susceptibility test (AST) devices that are used to determine minimum inhibitory concentrations (MIC) and/or interpretive category determinations of susceptible, intermediate and resistant (SIR) strains of bacteria to antimicrobial agents in medical laboratories. ISO 20776-2:2007 specifies requirements for AST devices (including diffusion test systems) and procedures for assessing performance of such devices. It defines how a performance evaluation of an AST device is to be conducted. ISO 20776-2:2007 has been developed to guide manufacturers in the conduct of performance evaluation studies.
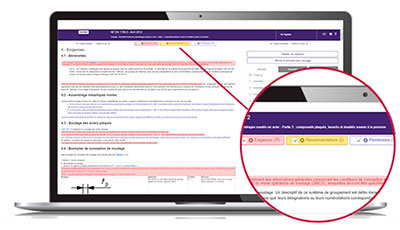
The Requirements department helps you quickly locate within the normative text:
- mandatory clauses to satisfy,
- non-essential but useful clauses to know, such as permissions and recommendations.
The identification of these types of clauses is based on the document “ISO / IEC Directives, Part 2 - Principles and rules of structure and drafting of ISO documents ”as well as on a constantly enriched list of verbal forms.
With Requirements, quickly access the main part of the normative text!
At a glance, you will be able to identify the additions, deletions or modifications to a text, table, figure and formula.
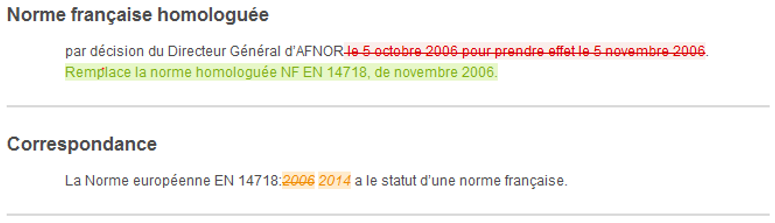
The Redlines + service is offered to you on the collection of French standards in force, in French language and in HTML and PDF format.
For an overview of the service, click on View a standard in redline format
COBAZ is the simple and effective solution to meet the normative needs related to your activity, in France and abroad.
Available by subscription, CObaz is THE modular solution to compose according to your needs today and tomorrow. Quickly discover CObaz!
Request your free, no-obligation live demo
I discover COBAZ