ISO 27269:2025
Health informatics - International patient summary
This document defines the core data set for a concise international patient summary (IPS), which supports continuity of care for a person and assists with coordination of their care.This document provides an abstract definition of a patient summary from which derived models are implementable.This document does not cover the workflow processes of data entry, data collection, data summarization, subsequent data presentation, assimilation, or aggregation. Furthermore, this document does not cover the summarization act itself, i.e. the intelligence, skills and competences, that results in the data summarization workflow. It is not an implementation guide that is concerned with the various technical layers beneath the application layer. Representation by various coding schemes, additional structures and terminologies are not part of this document.
This document defines the core data set for a concise international patient summary (IPS), which supports continuity of care for a person and assists with coordination of their care.
This document provides an abstract definition of a patient summary from which derived models are implementable.
This document does not cover the workflow processes of data entry, data collection, data summarization, subsequent data presentation, assimilation, or aggregation. Furthermore, this document does not cover the summarization act itself, i.e. the intelligence, skills and competences, that results in the data summarization workflow. It is not an implementation guide that is concerned with the various technical layers beneath the application layer. Representation by various coding schemes, additional structures and terminologies are not part of this document.
This document defines the core data set for a patient summary document that supports continuity of care for a person and coordination of their healthcare. It is specifically aimed at supporting the use case? scenario for ?unplanned, cross border care? and is intended to be an international patient summary (IPS). Whilst the data set is minimal and non-exhaustive, it provides a robust, well-defined core set of data items. The tight focus on this use case also enables the IPS to be used in planned care. This means that both unplanned and planned care can be supported by this data set within local and national contexts, thereby increasing its utility and value. It uses the European Guideline from the eHN as the initial source for the patient summary requirements, then takes into consideration other international patient summary projects to provide an interoperable data set specification that has global application. This document provides an abstract definition of a Patient Summary from which derived models are implementable. Due to its nature therefore, readers should be aware that the compliance with this document does not imply automatic technical interoperability; this result, enabled by this document, can be reached with the conformity to standards indicated in the associated technical specification and implementation guides. This document does not cover the workflow processes of data entry, data collection, data summarization, subsequent data presentation, assimilation, or aggregation. Furthermore, this document does not cover the summarization act itself, i.e. the intelligence/skill/competence that results in the data summarization workflow. It is not an implementation guide that is concerned with the various technical layers beneath the application layer. Implementation guidance for specifically jurisdictional concerns, e.g. Directives, terminologies, formats, etc., an example is specified in the associated Technical Specification[3]. In particular, representation by various coding schemes, additional structures and terminologies are not part of this document. Terminology and its binding are addressed in Reference [3]. The Identification of Medicinal Products standards (abbreviated to IDMP) are the recommended target for the Medication Summary related to this document but, prior to IDMP?s full implementation in practice, this IPS standard cannot insist in its use at this point in time and recognizes that interim schemes might be necessary until IDMP becomes established as a norm.
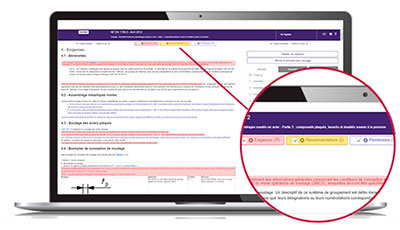
The Requirements department helps you quickly locate within the normative text:
- mandatory clauses to satisfy,
- non-essential but useful clauses to know, such as permissions and recommendations.
The identification of these types of clauses is based on the document “ISO / IEC Directives, Part 2 - Principles and rules of structure and drafting of ISO documents ”as well as on a constantly enriched list of verbal forms.
With Requirements, quickly access the main part of the normative text!
At a glance, you will be able to identify the additions, deletions or modifications to a text, table, figure and formula.
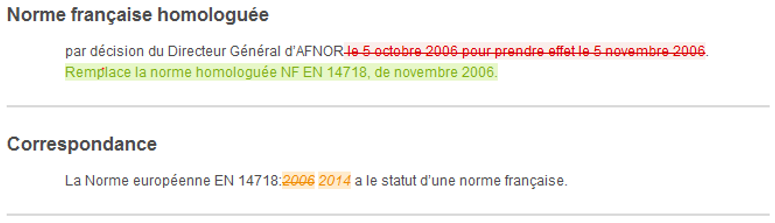
The Redlines + service is offered to you on the collection of French standards in force, in French language and in HTML and PDF format.
For an overview of the service, click on View a standard in redline format
COBAZ is the simple and effective solution to meet the normative needs related to your activity, in France and abroad.
Available by subscription, CObaz is THE modular solution to compose according to your needs today and tomorrow. Quickly discover CObaz!
Request your free, no-obligation live demo
I discover COBAZ